Definition
a thick, wet, unpleasant substance that covers something
Slime is made from PVA school glue and a natural mineral called Borax, which is found in household items, such as eye contact solution and natural laundry detergents. When mixed, a reaction forms between the ions in Borax with the molecules in the PVA glue occurs, creating slime. We also use other safe ingredients to create different textured slimes, such as instant snow or biodegradable beads to create a unique experience with each slime.

Despite both of these products being safe to play with, slime should never be consumed. We also do not recommend people with highly sensitive skin or allergies to household items to play with slime, despite never having any complaints about such issues.
Origin and usage
The word slime likely comes from the Old English word ‘slim’ which was probably related to the Old English ‘lim’ meaning ‘sticky substance’. Slime is related to words in many other languages, including the Dutch word ‘slijm’ meaning ‘phlegm’, the German ‘Schleim’ (‘slime’), and the Latin ‘limus’ meaning ‘mud or mire’.
Difference: Fluid and Slime
Liquids consist of many small particles that are densely packed together but can move freely against each other. In thin liquids, the particles are usually small and move past each other quite unhindered. In viscous liquids, the particles are larger and often contain long chains that intertwine.
A good slime is wobbly, can be kneaded, formed, torn, and adheres easily to surfaces such as fingers without leaving any residue. It is a gel. A gel consists of at least two components: One component is solid and forms a 3D network. The other component is a liquid or gas that fills the pores of the network.
Original Slime
The original slime or DIY slime contains polyvinyl alcohol (PVA), borax (sodium tetraborate), and water. If borax is dissolved in water, boric acid is formed, which reacts further to tetrahydroxyborate. Tetrahydroxyborate reacts with PVA. The OH groups of both particles combine to form solid compounds so that the borate anions become “nodes” in a flexible network. In addition, water is formed which is stored in the pores of the network.
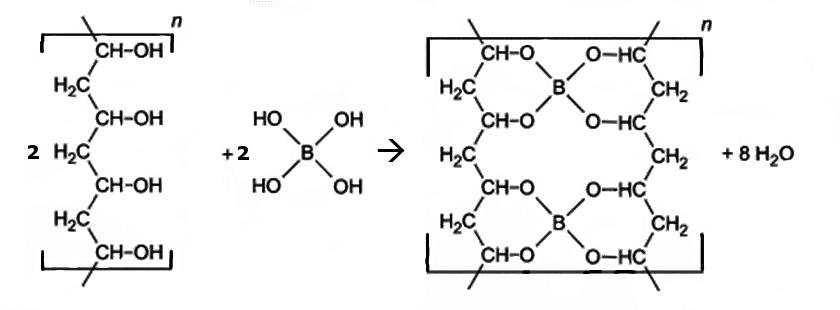
The bonds in the PVA borate network can be broken and newly formed relatively easily. Thus, the network is very changeable during kneading. This makes the slime really slimy.
Disposal
Borax and other boric acid compounds can damage unborn babies in the womb and impair fertility. Therefore, they are no longer sold to private households in EU countries and are banned from school laboratories. Borax must be disposed of as hazardous waste. If slime residues contain borax, they should be disposed of as hazardous waste.
How to make slime?
Though there are many variations, play slime can be made with a few basic ingredients and a simple recipe:
• Two 120ml bottles of liquid PVA glue
• Food colouring or glitter (optional)
• 5ml bicarbonate of soda
• 45ml saline solution (used for contact lenses)
To make slime, simply mix the ingredients in order in a large bowl. Continue stirring, then knead the mixture until it becomes thick and smooth. Slime can be stored for several weeks in an airtight container kept at room temperature.
Once you have the basic recipe perfected, you can experiment with your own slime variations by mixing colours and adding things like plastic gems, sequins or metallic confetti. Making slime is a great way to get kids busy in the kitchen, helping them practise important skills like measuring. Playing with slime is a fun, hands-on activity that can keep children occupied for hours!


You must be logged in to post a comment.