
“The unthinkable is that we’re distorting this atmospheric balance. We’re shifting the chemical balance so that we have more poisons in the atmosphere – ozones and acid rain on ground level – while we’re also changing the thermal climate of the earth through the greenhouse effect and – get this – simultaneously causing destruction of our primary filter of ultraviolet light. It’s incredible. Talk about the national-debt crisis – we’re piling up debts in the atmosphere, and the piper will want to be paid.”
~ Michael Oppenheimer
Introduction
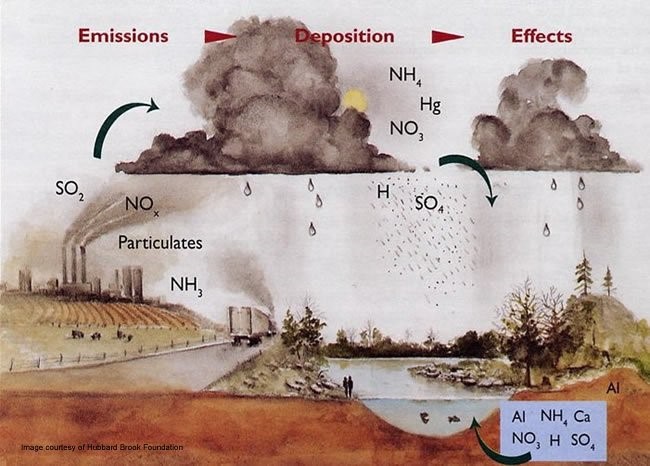
Acid rain, or acid deposition, is a broad term that includes any form of precipitation with acidic components, such as sulfuric or nitric acid that fall to the ground from the atmosphere in wet or dry forms. This can include rain, snow, fog, hail or even dust that is acidic.

Acid rain is caused by a chemical reaction that begins when compounds like sulfur dioxide and nitrogen oxides are released into the air. These substances can rise very high into the atmosphere, where they mix and react with water, oxygen, and other chemicals to form more acidic pollutants, known as acid rain.
It can have harmful effects on plants, aquatic animals, and infrastructure.
Effects of Acid Rain
The ecological effects of acid rain are most clearly seen in aquatic environments, such as streams, lakes, and marshes where it can be harmful to fish and other wildlife. As it flows through the soil, acidic rain water can leach aluminum from soil clay particles and then flow into streams and lakes.

Acid rain has been shown to have adverse impacts on forests, freshwaters, and soils, killing insect and aquatic life-forms, causing paint to peel, corrosion of steel structures such as bridges, and weathering of stone buildings and statues as well as having impacts on human health.
Acid rain also causes the corrosion of water pipes. Which further results in leaching of heavy metals such as iron, lead and copper into drinking water.
Prevention
1. The only precaution that we can take against acid rain is having a check at the emission of oxides of nitrogen and sulphur.
2. We have so far seen the details of acid rain and its harmful effect on animals, plants and the monuments.
3. Being responsible citizens, one should be aware of the harmful effects they cause and of the industries which give out nitrogen and sulphur compound wastes unethically. 4.A great way to reduce acid rain is to produce energy without using fossil fuels. Instead, people can use renewable energy sources, such as solar and wind power. Renewable energy sources help reduce acid rain because they produce much less pollution.
Acid Rain in India
Analysis of rainwater samples from Nagpur, Mohanbari (in Assam), Allahabad, Visakhapatnam and Kodaikanal in the decade 2001-2012 showed a pH level varying from 4.77 to 5.32, indicating that these places have actually been receiving ‘acid rain’. Rainwater with pH below 5.65 is considered acidic.
As energy requirements in India will grow rapidly in tune with the economy, coal dependence is expected to increase threefold over the current level of consumption, making the clouds of acid rain heavier over many highly sensitive areas in the country – the northeast, parts of Bihar, Orissa, West Bengal ,etc ,.

The rains in the Indian Ocean and the Arabian Sea and the Bay of Bengal have become acidic. Studies show the importance to regularly monitor more places for acid rains. But the mechanism to study acid rains is at present inadequate in India. imd stations are not located in the most polluted areas in the country.
Link


You must be logged in to post a comment.