Citation
Okeke, N. C., Chukwuemeka, G. N., Ubaezuonu, C. A., & Okocha, O. G. (2026). Public Perception of Factors Associated with Antenatal Care Utilization among Women in Awka South LGA of Anamabra State, Nigeria. Think India Quarterly, 29(1), 106–125. https://doi.org/10.26643/think/4
1Ngozi Chinenye Okeke, 2Gloria Nwakego Chukwuemeka, 2Chisom Anaestasi Ubaezuonu and 1Onyedikachi Goodness Okocha
cng.okeke@unizik.edu.ng; gn.chukwuemeka@unizik.edu.ng; ca.onwuamaeze@unizik.edu.ng;
1Department of Sociology, 2Department of Mass Communication
Nnamdi Azikiwe University, Awka, Anambra State.
Corresponding author: Ngozi Chinenye Okeke, Department of Sociology, Nnamdi Azikiwe University, Awka, Anambra, Nigeria. Email: cng.okeke@unizik.edu.ng. ORCID number: 0000-0001-6636-5166
Abstract
This paper examined public perception of the factors associated with antenatal care utilization among women in Awka South LGA. The objectives of this study were guided by conceptual issues such as; the state of antenatal care utilization among women, factors influencing antenatal care utilization among women, effects of poor antenatal care utilization among women, measures that could be put in place to improve antenatal care utilization among women in Awka South LGA. The research employed a mixed methods research design using a sample size of 204 respondents. Data collected from the questionnaire were processed using the Statistical Package for the Social Science (SPSS) software application version 23. The data were analyzed using descriptive statistics such as frequency tables and simple percentages. The hypotheses were tested using the Chi-square () statistics. The qualitative data collected from the field were transcribed which was thoroughly edited, analyzed thematically using narrative method of qualitative data analysis. The study identified distance to clinic as primary socio-cultural factors that determine the utilization of antenatal care service among women in Awka South Local Government Area. The study also found out that maternal death, pregnancy complications, infant death among others as effects of poor antenatal care services in Awka South Local Government Area. Finally, the research recommends that free and subsidized antenatal care service, improved facility infrastructure, community mobilization among others were listed as measures to improve antenatal care utilization among women in Awka South LGA.
Keywords: Antenatal care service, Antenatal care utilization, and Socio-economic factors
Introduction
Antenatal care (ANC) is the healthcare provided to women who are pregnant, for confirmation and monitoring of the progress of their pregnancy, and to promote their birth preparedness and complication readiness for ensuring optimal birth outcomes for both the mother and her baby (Marie, et. Al., 2022). Timely and quality antenatal care is a crucial determinant towards the prevention of maternal mortality, which is a significant developmental goal for developing countries, which contributes to more than 99% of maternal deaths worldwide (WHO, 2025).
Globally, ANC utilization has been recognized as an essential component of maternal healthcare, and efforts have been made to improve ANC utilization rates (World Health Organization, 2016). However, ANC utilization remains suboptimal in many low- and middle-income countries, including Nigeria (Federal Ministry of Health, 2013). According to the World Health Organization (2016), the ANC utilization rate in low- and middle-income countries is lower than the recommended standard. In Nigeria, the ANC utilization rate is lower than the World Health Organization’s recommended standard of at least four ANC visits per pregnancy (World Health Organization, 2016). According to the National Demographic and Health Survey (2018), only 58% of pregnant women in Nigeria attend ANC at least four times during their pregnancy. This low ANC utilization rate is attributed to various factors, including sociocultural, economic, and healthcare system-related factors.
Sociocultural factors, such as cultural beliefs, religious beliefs and practices, play a significant role in influencing ANC utilization among women in Nigeria. For instance, some women in Nigeria believe that pregnancy is a normal process that does not require medical attention, leading to low ANC utilization (Oladapo et al., 2019). Additionally, some cultural practices, such as the preference for traditional birth attendants, hinder ANC utilization among women in Nigeria (Egondi et al., 2017). Social factors, such as poverty and lack of access to healthcare facilities, are also significant barriers to ANC utilization among women in Nigeria. Many women in Nigeria cannot afford the cost of ANC services, leading to low ANC utilization (Adekanle et al., 2017). Furthermore, the lack of access to healthcare facilities, particularly in rural areas, hinders ANC utilization among women in Nigeria (Egondi et al., 2017). Healthcare system-related factors, such as the availability and quality of ANC services, also influence ANC utilization among women in Nigeria. The shortage of skilled healthcare providers, particularly in rural areas, hinders ANC utilization among women in Nigeria (Egondi et al., 2017). Additionally, the poor quality of ANC services, including the lack of essential equipment and supplies, deters women from utilizing ANC services (Adekanle et al., 2017). Lack of education further affects ANC use significantly as those without proper education are unable to see the benefits of ANC use.
In Anambra State the ANC utilization rate is lower than the national average. According to the Anambra State Ministry of Health (2020), only 50% of pregnant women in Anambra State attend ANC at least four times during their pregnancy. This low ANC utilization rate is attributed to various factors, including sociocultural, economic, and healthcare system-related factors. Awka South LGA is one of the areas in Anambra State where ANC utilization is a concern. The LGA has a population of over 200,000 people, with a significant proportion being women of reproductive age (National Population Commission, 2016). However, the ANC utilization rate in Awka South LGA is lower than the state average, with only 45% of pregnant women attending ANC at least four times during their pregnancy (Anambra State Ministry of Health, 2020). Therefore, there is a need to explore the public perception of the factors influencing ANC utilization among women in Awka South LGA.
Research Questions
The following research questions are put forward to guide the study
- What is the state of antenatal care utilization among women in Awka South LGA?
- What are the factors influencing antenatal care utilization among women in Awka South LGA?
- What are the effects of poor antenatal care utilization among women in Awka South LGA?
- What measures could be put in place to improve antenatal care utilization among women in Awka South LGA?
Study Hypotheses
The following hypotheses were formulated to guide this study
- Women with higher levels of education are more likely to utilize antenatal care than women with lower levels of education
- Women from low-income families are less likely to access antenatal care than their counterparts from high income families.
Research Methodology
The study adopted a mixed method research design, integrating quantitative and qualitative approach to provide a comprehensive understanding of public perception of the factors influencing antenatal care utilization among women. The study was conducted in Awka South Local Government Area of Anambra State, Nigeria, comprising nine towns and serving as the administrative center of the state. The target population include women of reproductive age (15years and above). The estimated target population was based on health records from local primary health centers, which indicated over 3,000 women fitting this description across the nine towns in the LGA. The scope of the study was limited to examining public perceptions of the state, influencing factors, effects, and improvement strategies regarding antenatal care utilization among women in the area. A sample size of 204 respondents was determined using Taro Ymane’s formula, while multi-stage sampling technique involving cluster, random and systematic sampling was used to select participants. Data were collected using structured questionnaire and in-depth interview guides. Questionnaires were administered face to face with the support of trained research assistants, while four purposely selected participants took part in in-depth interviews. Qualitative data were analyzed using SPSS with descriptive statistics and chi-square tests, while qualitative data were analyzed thematically and used to complement the quantitative findings.
Results
In this study, 204 questionnaires were administered by the researcher, out of which 198 (96.56%) of the questionnaires were correctly filled and returned. Six questionnaires were not completely filled. The analysis is consequently based on the correctly filled and returned 198 questionnaires. The quantitative data were also complemented by data from the in-depth-interview.
Socio-demographic Data of Respondents
Table 1: Distribution of Respondents by their Socio-Demographic Characteristics
| Social Demo-graphic Variables Responses | Frequency | Percent | |
| Age | 15-24 | 144 | 72.7 |
| 25 -34 | 39 | 19.7 | |
| 35- 43 44 and above | 8 7 | 4 3.5 | |
| Total | 198 | 100.0 | |
| Sex(Gender) | Male | 0 | 0 |
| Female | 198 | 100.0 | |
| Total | 198 | 100.0 | |
| Religious Affiliation | Christianity | 194 | 98 |
| Islam | 1 | 0.5 | |
| Traditional African religion | 3 | 1.5 | |
| Subtotal | 198 | 100 | |
| Total | 198 | 100.0 | |
| Education Qualification | No Formal Education | 0 | 0 |
| Primary | 25 | 12.6 | |
| Secondary Tertiary Post-Graduates | 114 19 40 | 57.6 9.6 20.2 | |
| Total | 198 | 100.0 | |
| Marital Status | Single | 192 | 97 |
| Married | 3 | 3 | |
| Divorced Separated Widowed | 0 0 0 | 0 0 0 | |
| Total | 198 | 100.0 | |
| Occupation | Unemployed | 86 | 43.4 |
| Civil Servant | 6 | 3.5 | |
| Trader Artisan Student Total | 5 1 101 198 | 2.5 0.5 51 100.0 | |
| Income | 1000- 30000 | 133 | 67.1 |
| 31000 – 60000 | 34 | 17.1 | |
| 61000 – 90000 | 5 | 2.5 | |
| 91000 and above | 26 | 13.3 | |
| Total | 198 | 100.0 | |
Field survey 2025
Table 1 show that 144(72.2%) constituting the majority of the respondents are within the age bracket of 15-24 years. The mean age of respondents is 19.5 years old. It could be seen also that Females are the gender of the study. 194(98%) of the respondents are Christians, 1(0.5%) respondent are Islam while 3(1.5%) respondents are of the Africa Traditional Religion. Similarly, 25(12.6%) respondents have primary certificate as the highest education qualification, 114(57.6%) respondents have secondary certificate, 9(9.6%) respondents have tertiary certificate, while 40(20.2%) respondents had Post-Graduate Degree. Also, 86(43.4%) of the respondents are unemployment, 6(3.5%) respondents are civil servant, 5(0.5%) respondents identified as trader, while 101(51%) respondents are unemployed. However, 133(67.2%) of the respondents earn s between #1000- #30000 monthly, 34(17.2%) respondents earn #31000 – #60000 monthly, 5(2.5%) respondents earn #61000 – #90000, while 26(13.3) earns #91000 and above. Table1also shows that majority of the respondents are single, 6(3%) are married, while there were no responses for, separated, divorced or widowed.
Substantive Issues of the Research
This section deals with the analysis of research question formulated to guide the study
Research Question One: What is the state of antenatal care utilization among women in Awka South LGA? To answer the research question, responses to research questionnaire 8 to 11 were analyzed on table and chart below.
Table 2: Distribution of respondents’ views on the awareness of the importance of antenatal care services
| Responses | Frequency | Percent |
| Yes | 198 | 100 |
| No | 0 | 0 |
| Total | 198 | 100 |
Field survey 2025
Table 2 shows that 198 (100%) respondents held the view that they are aware of the importance of antenatal care services in Awka South LGA. Thus, all the respondents held the view that they are aware of the importance of antenatal care services in Awka South LGA.
Table 3: Distribution of respondents’ views on whether they have attended antenatal care during pregnancy
| Responses | Frequency | Percent |
| Yes | 79 | 39.9 |
| No | 119 | 60.1 |
| Total | 198 | 100 |
Field survey 2025
The information on the above table shows that 119(60.1%) respondents held the view that they haven’t attended antenatal care services during pregnancy in Awka South LGA, while 79(39.9%) of the respondents indicated that they have attended antenatal during. Summarily, majority (60.1%) of the respondents were of the view that they haven’t attended antenatal care services during pregnancy in Awka South LGA.
Table 4: Distribution of respondents’ views on number of times they have attended antenatal care during pregnancy
| Responses | Frequency | Percent |
| Once | 10 | 12.6 |
| Twice | 4 | 5.1 |
| Three times | 6 | 7.6 |
| Four times or more | 59 | 74.7 |
| Total | 79 | 100.0 |
Field survey 2025
Table 4 shows that 59(74.7%) of respondents held the view that they attended antenatal care services four times and more, while 4(5.1%) of the respondents indicated that they attended antenatal care services once in Awka South LGA. This implies that majority (74.7%) of the respondents opted that they attended antenatal care services four times and more in Awka South LGA
Table 5: Distribution of respondents’ views on where they usually attend antenatal care
| Responses | Frequency | Percent |
| Government hospital Private clinic Traditional birth attendant Church/ Mosque-based center | 129 56 0 13 | 65.1 28.3 0 6.6 |
| Total | 198 | 100.0 |
Field survey 2025
Table 5 shows that 129(65.1%) of the respondents were of the view that they usually attend antenatal at government hospital in Awka South LGA, while 13(6.6%) of the respondents indicated church/ mosques-based centre in Awka South LGA. Thus, majority (65.1%) of the respondents indicated that they usually attend antenatal at government hospital in Awka South LGA.
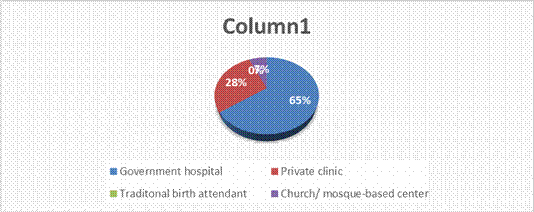
Figure One. Distribution of respondents’ views on where they usually attend antenatal care
Research Question Two: What are the factors influencing antenatal care utilization among women in Awka South LGA? To answer the research question, responses to research questionnaire 12 to 19 were analyzed on table and chart below.
Table 6: Distribution of respondents’ views on major factors that influence their decision to attend antenatal care
| Responses | Frequency | Percent |
| Cost of service Distance to clinic Husband’s support Health worker attitude Lack of Knowledge | 42 83 7 66 0 | 21.2 42 3.5 33.3 0 |
| Total | 198 | 100 |
Field survey 2026
Table 6 shows that 83(42%) of the respondents held the view that distance to clinic influence their decision to attend care in Awka South LGA, while 7(3.5%) of the respondents chose husband support. Summarily, majority (42%) of the respondents were of the view that distance to clinic influence their decision to attend care in Awka South LGA; response from the qualitative instrument state
…….In my opinion, I think what mostly influences the decision to attended antenatal care is the partners’ support and financial cost of the services. Most pregnancy women seldom attend antenatal care until they 7-8month pregnant which is very wrong and when asked, they either make reference to their partner or cost(IDI, 26year, Female, Nurse, Single).
Table 7: Distribution of respondents’ views on whether cultural belief discourages the use of antenatal services
| Responses | Frequency | Percent |
| Yes | 132 | 66.8 |
| No | 66 | 33.3 |
| Total | 198 | 100 |
Field survey 2025
Table 7 shows that 132 (66.8%) of the respondents held the view that cultural belief discourages the use of antenatal in Awka South LGA, while 66(33.3%) respondents indicated that it doesn’t. Thus, majority (66.8%) of the respondents were of the view that cultural belief discourages the use of antenatal in Awka South LGA.
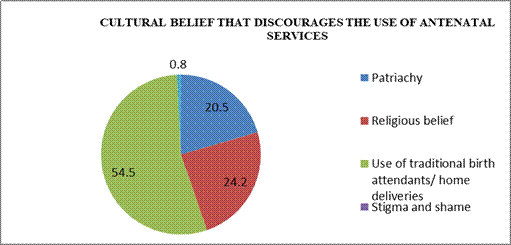
Field survey 2025
Figure Two: Distribution of respondents’ views on the cultural belief that discourages the use of antenatal services
Figure 2 illustrate that 72(54.5%) of respondents held the view that use of traditional birth attendants/ home deliveries discourages the use of antenatal services, while 1(0.8%) of the respondents indicated fear. Thus, majority (54.5%) of the respondents were of the view that use of traditional birth attendants/ home deliveries discourages the use of antenatal services in Awka South LGA.
Table 8: Distribution of respondents’ views on whether level of education affects decision to use antenatal services
| Responses | Frequency | Percent |
| Yes | 198 | 100 |
| No | 0 | 0 |
| Total | 198 | 100 |
Field survey 2025
Table 8 shows that 198 (66.8%) of the respondents held the view that level of education affects decision to use antenatal care in Awka South LGA. Thus, majority (100%) of the respondents were of the view that level of education affects decision to use antenatal care in Awka South LGA.
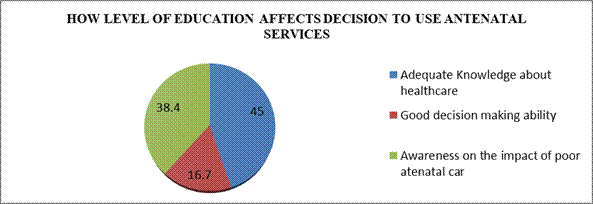
Field survey 2025
Figure Three: Distribution of respondents’ views on how level of education affects decision to use antenatal services
Figure 3 illustrate that 89(45%) of respondents held the view that academic knowledge about healthcare affect decision on the use of antenatal services, while 33(16.7%) of the respondent good decision-making ability. Thus, majority (45%) of the respondents were of the view that academic knowledge about healthcare affect decision on the use of antenatal services in Awka South LGA.
Table 9: Distribution of respondents’ views on whether employment status affects women’s antenatal care utilization
| Responses | Frequency | Percent |
| Yes | 198 | 100 |
| No | 0 | 0 |
| Total | 198 | 100 |
Field survey 2025
Table 9 shows that 198 (66.8%) of the respondents held the view that employment status affects women’s antenatal care utilization in Awka South LGA. Thus, majority (100%) of the respondents were of the view that employment status affects women’s antenatal care utilization in Awka South LGA.
Table 10: Distribution of respondents’ views on whether monthly income influences the use of antenatal care services
| Responses | Frequency | Percent |
| Yes | 198 | 100 |
| No | 0 | 0 |
| Total | 198 | 100 |
Field survey 2025
Table 10 shows that 198 (66.8%) of the respondents held the view that monthly income influence the use of antenatal care service in Awka South LGA. Thus, majority (100%) of the respondents were of the view that monthly income influences the use of antenatal care service in Awka South LGA. Responses from the qualitative data states
…..It is very crystal clear that the level of wealth one has, have a long way of determining his/her lifestyle; so does it in health. You can’t speak of health insurance or antenatal care when you barely manage to feed or doing a diagnoses before buying medics for ailment. What am saying is that no matter your level of education or knowledge in health, once there’s no financial backing, you are going nowhere (IDI, 32year, Male, Health personnel, Married)
Research Question Three: What are the effects of poor antenatal care utilization among women in Awka South LGA? To answer the research question, responses to research questionnaire 20 and 21 were analyzed on table and chart below.
Table 11: Distribution of respondents’ views on the consequences of not attending antenatal care
| Responses | Frequency | Percent |
| Maternal death Infant death | 0 6 | 0 3 |
| Pregnancy complications All of the above | 46 114 | 23.2 73.7 |
| Total | 198 | 100.0 |
Field survey 2025
Table 11 shows that 114(73.7%) of respondents held the view that all of the above mention options are the consequences of not attending antenatal care in Awka South LGA, while 6(3%) of the respondents indicated infant death. Thus majority (73.7%) of the respondents were of the view that all of the above mention options are the consequences of not attending antenatal care in Awka South LGA; Responses from the qualitative data states;
…..there are consequences for everything which is vital but neglected, so it is for antenatal care for every pregnant women. There may be factors that might have prevented the utilization of antenatal care services but the effects are there and one of the numerous effects is health complications of the pregnant woman (IDI, 32year, Male, Health personnel, Married)
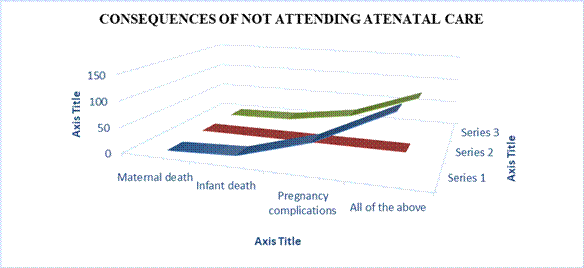
Figure Four. Distribution of respondents’ views on the consequences of not attending antenatal care
Table 12: Distribution of respondents’ views on whether they know any woman who have
experienced complications due to poor antenatal attendance
| Responses | Frequency | Percent |
| Yes | 33 | 16.7 |
| No | 165 | 83.3 |
| Total | 198 | 100 |
Field survey 2025
Table 12 shows that 165 (83.3%) of the respondents held the view that they don’t any woman who have experienced complications due to poor antenatal attendance, while 33(16.7%) of the respondents indicated that they have. Thus, majority (83.3%) of the respondents were of the view that they don’t any woman who have experienced complications due to poor antenatal attendance in Awka South LGA.
Research Question Four: What measures could be put in place to improve antenatal care utilization among women in Awka South LGA? To answer the research question, responses to research questionnaire 22 to 24 were analyzed on table and chart below
Table 13: Distribution of respondents’ views on what can be done to improve antenatal care utilization
| Responses | Frequency | Percent |
| Health education and sensitization | 13 | 6.6 |
| Free or subsidized services | 97 | 49 |
| Community mobilization Improved facilities infrastructure | 78 10 | 40.4 5.1 |
| Total | 198 | 100.0 |
Field survey 2025
Table 13 shows that 97(49%) respondents held the view that free or subsidized antenatal services can help improve antenatal care utility in Awka South LGA, while 13(6.6%) of the respondents indicated health education and sensitization. Thus, majority (49%) of the respondents were of the view that free or subsidized antenatal services can help improve antenatal care utility in Awka South LGA.
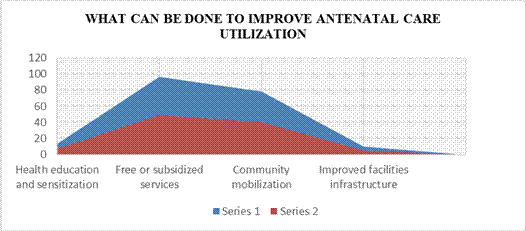
Figure Five: Distribution of respondents’ views on what can be done to improve antenatal care utilization
Table 14: Distribution of respondents’ views on whether government and NGOs should collaborate to improve antenatal care service
| Responses | Frequency | Percent |
| Yes | 198 | 100 |
| No | 0 | 0 |
| Total | 198 | 100 |
Field survey 2025
Table 14 shows that 198 (100%) respondents held the view that government and NGOs should collaborate to improve antenatal care service in Awka South LGA. Thus, all (100%) the respondents held the view that government and NGOs should collaborate to improve antenatal care service in Awka South LGA. Responses from the qualitative data states;
…….After God na government, I think the government are in the best position to improve the situation. Government self, na man dominant; but honestly, there are policies that could help to dicey the situation so as to improve antennal services. If not for one thing, at least the health sector knowing full well that women are the mechanism for population or reproduction. The government should make provision of fuctional primary healthcare centers accessible to every woman (IDI, 22year, female, Health personnel, Single).
Table 15: Distribution of respondents’ views on whether social workers and community leaders can play a role in encouraging antenatal care attendance
| Responses | Frequency | Percent |
| Yes | 198 | 100 |
| No | 0 | 0 |
| Total | 198 | 100 |
Field survey 2025
Table 15 shows that 198 (100%) of the respondents held the view that social workers and community leaders can play a role in encouraging antenatal care attendance. Thus, all (100%) of the respondents were of the view that social workers and community leaders can play a role in encouraging antenatal care attendance in Awka South LGA.
Test of Study Hypotheses
Details of test of the two hypotheses postulated for this study are shown hereunder:
Study Hypothesis One: Women with higher levels of education are more likely to utilize antenatal care than women with lower levels of education. Data on table 1 and 5 formed the basis for testing hypothesis one.
Table 16: Relationship between level of education and utilize antenatal care service in Awka South LGA
| Educational Qualification | Distribution of respondents’ views on where they usually attend antenatal care | ||||
| Government Hospital | Private Clinic | Traditional birth attendants | Church/ Mosque-Based center | Total | |
| No formal Education | 0 | 0 | 0 | 0 | 0 |
| Primary | 19 | 4 | 0 | 2 | 25 |
| Secondary | 84 | 24 | 0 | 6 | 114 |
| Tertiary | 9 | 7 | 0 | 3 | 19 |
| Post-Graduates | 17 | 21 | 0 | 2 | 40 |
| Total X2=24.53,DF=12,Pvalue=0.001 | 129 | 56 | 0 | 13 | 198 |
Source: SPSS version 25
The computed value of the chi-square is 24.53 while the table value of chi-square at 0.05level of significance with a degree of freedom (DF) of 12 is 21.026. Since the computed value of chi-square is greater than the table value, the researcher accepted the alternative hypothesis. There is therefore a significant relationship between levels of education and utilize antenatal care in Awka South LGA.
Study Hypothesis Two: Women from low-income families are less likely to access antenatal care than their counterparts from high income families. Data on table 1 and 4 formed the basis for testing hypothesis two.
Table 17: Relationship between level of income and utilize antenatal care service in Awka South LGA
| Level of Income | Distribution of respondents’ views on where they usually attend antenatal care | ||||
| Government Hospital | Private Clinic | Traditional birth attendants | Church/ Mosque-Based center | Total | |
| 1000 – 30000 | 82 | 46 | 0 | 5 | 133 |
| 31000 – 60000 | 26 | 6 | 0 | 2 | 34 |
| 61000 – 90000 | 3 | 1 | 0 | 1 | 5 |
| 91000 and above | 18 | 3 | 0 | 5 | 26 |
| Total X2=22.1,DF=9,Pvalue=0.001 | 129 | 56 | 0 | 13 | 198 |
Source: SPSS version 25
The computed value of the chi-square is 22.1 while the table value of chi-square at 0.05level of significance with a degree of freedom (DF) of 9 is 16.919. Since the computed value of chi-square is greater than the table value, the researcher accepted the alternative hypothesis. There is therefore a significant relationship between levels of income and utilize antenatal care in Awka South LGA.
Discussion of Findings
This paper investigated the factors influencing antenatal care utilization among women in Awka South LGA. Two hundred and four (204) respondents were the study population within ages ranging from 15years and above. Two hypotheses were tested; hypothesis one stated that women with higher levels of education are more likely to utilize antenatal care than women with lower levels of education, while hypothesis two stated that women from low-income families are less likely to access antenatal care than their counterparts from high income families in Awka South LGA, Anambra State. Findings in all hypotheses tested shows that there is a significant relationship between both variables in Awka South LGA, Anambra State. From the quantitative data of the respondents, the study found out that majority of the respondents are male between the age of 15 to 24 and are single and of the Christian religious affiliation with secondary school certificate as the highest education attained by the majority of the study participants.
Findings from the analysis done on the responses to the questionnaire schedule; all of the respondents 198 (100%) indicated that they are aware of the importance of antenatal care service, while majority of the respondents indicated that they have attended antenatal at least four times or more during pregnancy. This is in consonance with the Anambra State Ministry of Health (2020), which noted that only 50% of pregnant women in Anambra State attend ANC at least four times during their pregnancy. This low ANC utilization rate is attributed to various factors, including sociocultural, economic, and healthcare system-related factors.
Findings in research question two, which set out to determine factors influencing antenatal care utilization in Awka South LGA; found out that majority 83(42%) of the respondents indicated distance to clinic, while others include cost of service, lack of knowledge amongst others. This is in consonance with A study by Ononokpono and Odimegwu (2014) found that Nigerian women with secondary education were twice as likely to attend ANC services compared to women without formal education. This correlation is attributed to greater awareness of the benefits of maternal healthcare and increased autonomy in decision-making. Similarly, financial stability plays a crucial role in ANC uptake, as out-of-pocket healthcare expenses can deter low-income women from seeking care (Basha, 2019).
Furthermore, findings in research question three, which set out to find the effects of poor antenatal care utilization in Awka South LGA; found out that it could lead to maternal death, infant death, pregnancy complications amongst others, however majority 114(73.7%) of the respondents chose all of the mentioned effects. This is in consonance with a study by Say et al. (2014), who stated that inadequate ANC is a major contributor to maternal deaths worldwide, particularly in low- and middle-income countries. The lack of routine check-ups prevents early detection of complications such as pre-eclampsia, gestational diabetes, and infections, which can be fatal if left untreated. According to Lawn et al. (2014), nearly 45% of neonatal deaths globally are attributed to complications arising from inadequate prenatal care.
The study further suggested measures to improve antenatal care utilization in Awka South LGA and these may include; collaboration between the government and NGOs in healthcare sector, health education and sensitization, improved facility infrastructure among others while a good number 97(49%) of the respondents indicated free and subsidized services which also is in consonance with Aiga et al. (2024) who noted that policy interventions and government commitment are fundamental to improving ANC utilization. Implementing and enforcing policies that mandate comprehensive maternal healthcare services, increasing healthcare funding, and integrating ANC services into primary healthcare systems ensure sustainable improvements in maternal health.
Finally, two hypotheses were formulated and tested. It was found that level of education can significantly influence the utilization of antenatal care among women in Awka South Local Government Area. Similarly, the study also found that there is a significant relationship level of income of female respondents and the utilization of antenatal care among women in Awka South L.G.A. This is in consonance with the social determinants of health theory by Marmot and Wilkinson (1999). The SDH posit that, it becomes clear that public perceptions are deeply embedded in socioeconomic and cultural realities. In many communities, especially in Nigeria, factors such as poverty, education level, and rural-urban disparities play a significant role in determining whether a woman seeks ANC services. Ononokpono and Odimegwu (2014) found that women with higher levels of education and greater financial stability were significantly more likely to attend at least four ANC visits compared to women with low income and little education. Public perception of ANC is shaped by these realities—when healthcare is seen as expensive or unnecessary, women are less likely to seek services
Conclusion
Based on the findings of the study, the researchers has been able to conclude that the phenomenon of antennal utilization among women is not new. The study further affirms that there are certain factors that determine the utilization of antenatal care service among women and the study also identified some of the measures to put in place to improve antenatal care. In this study two hypotheses were tested. Thus, the first hypothesis which stated that women with higher levels of education are more likely to utilize antenatal care than women with lower levels of education was confirmed and therefore accepted. The second hypothesis which state that women from low-income families are less likely to access antenatal care than their counterparts from high income families was also confirmed and therefore accepted. Based on the findings, some recommendations were made. Lastly, the study identified the need to address the problem and the role that can be played by different stake holders such as the social workers, community leaders, NGOs and the government in order to improve antenatal care utilization in the study.
Recommendations
Based on the research findings, the researchers put forward the following recommendations:
- Through the Ministry of woman affair: through the ministry of woman affair, policies to eliminate existing socio-cultural disparities and existing gender relations and power disparities between women and men. For instance, programmes aiming at improving women’s access to health care services by developing locally-based facilities should make sure that women-to-women services will be available, with opening times adapted to women’s needs and activities. This will help resolve the state of antenatal care utilization among women in Awka South LGA
- Mobilization of women’s organizations: women’s groups, unions, neighborhood associations and cooperatives in problem identification, strategy formulation and implementation. For instance, women’s organisations are likely to know which health problems women experience and which institutional, structural and cultural barriers impede their access to health care (e.g. in case of antenatal services). The will help checkmate the issue of patriarchy in resolving the factors influencing antenatal care utilization among women in Awka South LGA
- Health Education and Sensitization: For instance, instead of devaluating their knowledge, traditional healthcare providers could be targeted by skills training programmes. Their participation in the development of health strategies should be sought. These measures will help ameliorate the effects of poor antenatal care utilization among women in Awka South LGA
- Improved Facility Infrastructure; The government in collaboration with NGOs and stakeholder could join hand in improving our healthcare system and make it accessible and affordable for the low-income earners too because this strategy will improve antenatal care utilization among women in Awka South LGA
References
Adekanle, D. A., Isawumi, A. I., &Olayemi, O. O. (2017). Late antenatal care booking and its predictors among pregnant women in Nigeria. International Journal of Women’s Health, 9, 1–6. https://doi.org/10.2147/IJWH.S124766
Adepoju, A. O., Oladapo, O. T., &Iyaniwura, C. A. (2020). Barriers to antenatal care utilization in Nigeria: A qualitative study. BMC Pregnancy and Childbirth, 20(1), 1–10. https://doi.org/10.1186/s12884-020-03165-7
Aiga, H., Kawakatsu, Y., Kadoi, N., Obeng, E., Addai, F. T., Ofosu, F., Fujishima, K., Omachi, M., & Yamaguchi, E. (2024). Effectiveness of a community-based intervention package in maternal health service utilization: A cross-sectional quasi-experimental study in rural Ghana. PloS one, 19(11), e0311966. https://doi.org/10.1371/journal.pone.0311966
Anambra State Ministry of Health. (2020). ASMH antenatal care randomized trial: Manual for the implementation of the new model. ASMH.
Basha, G. W. (2019). Maternal healthcare utilization in Ethiopia: Trends and determinants. Ethiopian Journal of Health Development, 33(2), 1–8
Egondi, T., Oyolola, M., &Abuya, T. (2017). Cultural barriers to antenatal care in Nigeria. Journal of Biosocial Science, 49(4), 456–471. https://doi.org/10.1017/S0021932016000443
Federal Ministry of Health. (2013). National strategic health development plan framework (2010–2015). Federal Republic of Nigeria.
Izugbara, C. O., Wekesah, F. M., &Adedini, S. A. (2018). Male dominance and maternal healthcare in Nigeria. Culture, Health & Sexuality, 20(9), 981–995. https://doi.org/10.1080/13691058.2017.1407412
Lawn, J. E., Blencowe, H., Oza, S., You, D., Lee, A. C. C., Waiswa, P., Lalli, M., Bhutta, Z., Barros, A. J. D., Christian, P., Mathers, C., & Cousens, S. N. (2014). Every newborn: Progress, priorities, and potential beyond survival. The Lancet, 384(9938), 189–205. https://doi.org/10.1016/S0140-6736(14)60496-7
Marmot, M., & Wilkinson, R. G. (Eds.). (1999). Social determinants of health. Oxford University Press.
Marie, S., et al. (2022). Antenatal care and maternal outcomes in developing countries. Journal of Global Health, 12(1), 1–10.
Nigeria Demographic and Health Survey (NDHS) (2018). Abuja, Nigeria, and Rockville, Maryland, USA: NPC and ICF. http://ngfrepository.org.ng:8080/jspui/handle/123456789/3145
National Population Commission. (2016). Nigeria demographic and health survey 2018. NPC.
Oladapo, O. T., Iyaniwura, C. A., & Sule-Odu, A. O. (2019). Sociocultural determinants of antenatal care utilization in Nigeria. BMC Pregnancy and Childbirth, 19(1), 1–9. https://doi.org/10.1186/s12884-019-2220-z
Ononokpono, D. N., & Odimegwu, C. O. (2014). Determinants of maternal healthcare utilization in Nigeria. African Journal of Reproductive Health, 18(3), 130–137.
Say L, Chou D, Gemmill A, Tunçalp Ö, Moller AB, Daniels J, et al Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health. 2014;2(6):e323–33. doi: 10.1016/S2214-109X(14)70227-X
World Health Organization. (2002). WHO antenatal care randomized trial: Manual for the implementation of the new model. WHO.
World Health Organization. (2016). WHO recommendations on antenatal care for a positive pregnancy experience. WHO.
World Health Organization. (2025). Maternal Mortality. https://www.who.int/news-room/fact-sheets/detail/maternal-mortality




You must be logged in to post a comment.